Let’s start by alleviating the main concern: The content of the China-U.S. economic and trade agreement has no obvious negative impact on the business conducted in China among the foreign pharmaceutical and medical device companies we surveyed. This industry is particularly interested in intellectual property rights and trade expansion, including consideration of post-filing data, effective mechanisms for early resolution of patent disputes, patent term extensions, combating counterfeit drug manufacturing and information sharing, as well as expanding U.S. pharmaceutical products and medical equipment exports to China.
However, this part of the agreement still has no clear adverse effect on the industry. On the contrary, the importance of the protection of intellectual property rights and the increased importation of pharmaceutical products and medical devices will further provide opportunities and vitality for the development of pharmaceutical and medical device multinational corporations (MNCs) in the region.
China Remains a Key Market for Multinationals
No surveyed company had lowered their business expectations for the China market due to changes in China-U.S. relations, though some claimed to be impacted on export tariff and supply chain. Judging from perspectives of headquarter (HQ) controls, investment and expected growth, China remains the biggest market globally for these companies.
Among the 20 surveyed companies, seven claimed their general managers (GMs), or other function heads, in China report directly to the global HQ. Some China GMs are also members of global management. This indicates that business decisions in this region are communicated directly to the HQ with a high management level and frequency. Based on another survey we conducted in back in 2018, the closer an office is located to the global HQ, the better support it will receive, ultimately leading to improved business performance.
Meanwhile, our survey findings from this year indicated that 10 companies would raise investments in China, covering research and development (R&D), production, new product, digital transformation and external strategic cooperation. For example, from an R&D perspective, some companies plan to move R&D centers to China and accelerate production, bringing innovative drugs to market ahead of schedule and increasing budgets for clinical research in certain strategic therapeutic areas. When discussing external strategic cooperation, companies said they will look for more partners and increase investment in small business development projects to make up for the lack of production lines globally in the short term. Therefore, we anticipate more strategic partnerships between MNCs and local, private companies, such as Eli Lilly and Innovent, Pfizer and CStone, AstraZeneca and Yuwell, as well as Amgen and BeiGene.
Although various industries have been impacted by the COVID-19 pandemic around the world, the International Monetary Fund (IMF) predicted that China will be the only major economy to experience positive growth globally this year. Five surveyed companies said that China achieved the fastest recovery around the world. They expect the country to continue to enhance investment of new products and core therapeutic areas, while maintaining double-digit growth to make China the growth engine for global business.
Public statistics from the first half of 2020 show that leading pharmaceutical MNCs have maintained high growth in China compared to the same period last year, including 19% growth for Novartis, 14% for AstraZeneca and MSD, 12% for Novo Nordisk and 7% for Roche.
Enhancing Local Talent Developmment and Key Retention
Due to the recent changes in China-U.S. trade relations, companies said their employees or job candidates had concerns about their firms’ current and future situation. Further details revealed that they are facing difficulties in recruiting global talent, especially from the U.S., and transferring Chinese talent to the U.S. However, 80% said their talent attraction, retention and development were not impacted by the change. Faced with these challenges, firms will continue to develop local R&D and digital talent to support the growth and transformation of the China market. Many will also attract talent in government relations and strategic planning with better foresight to prepare for the future. The figure below shows the year-over-year voluntary, involuntary and overall turnover rate for pharmaceutical and medical device foreign invested companies in China. It is evident that the trend has remained mostly steady, increasing slightly over the past two years.
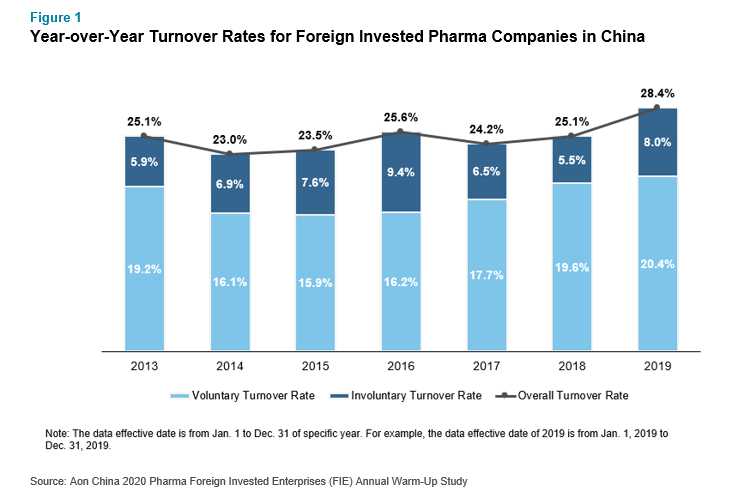
Major talent management issues and challenges for pharmaceutical and medical device MNCs in China come from policy change and local competition. For example, the accelerated approval of innovative medicine, the 2017 national medical insurance negotiation and the volume-based procurement policy of 2018, bring about both challenges and opportunities for business transformation, as well as new requirements on talent. The rise of local pharmaceutical and medical device companies with advanced technologies, strong capital investment and related policy support also enhances competition for talent.
Compared to developed MNCs, thriving local companies are more aggressive in talent attraction, offering higher pay, more incentives and additional resources. This gives an edge to local companies in talent attraction and retention. Therefore, MNCs should improve their talent development and retention strategies for business transformation and growth, allocate more resources to key talent and leverage their employee value proposition for career development and opportunities.
Next Steps
In general, surveyed company responses indicated that changes within China-U.S. relations have not had a significant impact on pharmaceutical and medical device MNCs in China to-date. The country remains a key market with great strategic importance and companies anticipate making additional investment in their business and talent in China.
Trade tensions between the two countries have not resulted in reduced trade volume. In fact, statistics from the General Administration of China Customs show that during the first nine months of the year, exports and imports between the two countries are up from 2019. Looking ahead, the impact from the changes in China-U.S. relations on non-sensitive industries and companies is expected to remain limited.
Pharmaceutical and medical device MNCs must continue to focus on successfully seizing opportunities from upcoming policies, the Healthy China 2030 Initiative and an aging population in China — all core influencing factors for business performance. The improvement of organizational capability, workforce agility and key talent attraction and retention will be essential for companies to adapt to rapid change, implement strategies and achieve better growth in the China market.
To learn more about these findings and the pharmaceutical and medical device industry in China, please reach out to Michelle Lv at
michelle.lv@aon.com. For additional information on participating in a survey or to speak with a member of our team, please write to
rewards-solutions@aon.com.